International Journal of Clinical and Medical Research | Year 2024 | Volume 1 | Issue 1 | Pages 31-38
Integration of Blood Brain Barrier: A Review Study
Shatha Ramadhan Zaidan 1*1Department of Microbiology, Alkindy College of Medicine, University of Baghdad, Baghdad, Iraq
View PDF Download XML Download DOI XML DOI: 10.66590/ijcmr2024010102
Abstract
Background: The blood-brain barrier (BBB) is a highly specialized structure that maintains brain homeostasis by regulating molecular exchange between the bloodstream and the central nervous system. Ischemic stroke disrupts BBB integrity, leading to edema, neuroinflammation, and poor outcomes. Understanding the genetic regulation of BBB components is critical for developing targeted therapies. Methods: This narrative review synthesizes current literature on BBB structure, the neurovascular unit, and genetic mechanisms underlying BBB dysfunction in ischemic stroke. Key genes involved in tight junctions, inflammatory responses, matrix remodeling, and angiogenesis were analyzed, with a focus on CLDN5, MMP-9, VEGF, and IL-6. Evidence from animal models (e.g., MCAO, knockout mice) and human genetic studies was evaluated. Results: Key findings show that CLDN5 downregulation increases BBB permeability and edema. Elevated MMP-9 degrades tight junction proteins and extracellular matrix, contributing to hemorrhagic transformation. VEGF overexpression promotes angiogenesis but exacerbates BBB leakage and edema. IL-6 drives neuroinflammation and correlates with worse stroke outcomes. Animal models confirm that knockout of MMP-9 or IL-6 reduces BBB disruption and infarct size, while CLDN5 knockout increases stroke susceptibility. Therapeutic perspectives include gene therapy (e.g., BDNF, VEGF modulation), CRISPR-based editing of MMP-9, and pharmacogenomics targeting ABCB1 and CYP2C19 variants. Conclusion: Genetic regulation of BBB integrity involves complex interactions among tight junction, inflammatory, and angiogenic genes. Targeting CLDN5, MMP-9, VEGF, and IL-6 pathways offers promising therapeutic avenues for ischemic stroke. Future research should focus on safe gene delivery and personalized medicine approaches.
INTRODUCTION
The blood-brain barrier (BBB) is a highly specialized structure that plays a critical role in maintaining brain homeostasis by regulating the exchange of molecules between the bloodstream and the central nervous system (CNS) [1]. Composed of endothelial cells, pericytes, astrocytes, and a basement membrane, the BBB ensures selective permeability, protecting the brain from harmful substances while allowing essential nutrients to pass through [2]. Disruption of BBB integrity is a hallmark of numerous neurological disorders, including ischemic stroke, which remains a leading cause of morbidity and mortality worldwide [3].
Ischemic stroke occurs due to the occlusion of cerebral blood vessels, leading to reduced blood flow, oxygen deprivation, and subsequent neuronal damage [4]. The BBB, which is normally impermeable to most blood-borne substances, becomes compromised during and after ischemic stroke, resulting in increased permeability, edema formation, and infiltration of inflammatory cells [5]. This disruption not only exacerbates brain injury but also complicates recovery, making BBB integrity a key focus of stroke research [6].
Emerging evidence suggests that genetic factors play a significant role in regulating BBB integrity both before and after ischemic stroke [7]. Genes encoding tight junction proteins, transporters, inflammatory mediators, and growth factors have been implicated in maintaining BBB stability and influencing stroke outcomes [8]. For instance, polymorphisms in genes such as CLDN5 (claudin-5) and MMP-9 (matrix metalloproteinase-9) have been associated with increased susceptibility to stroke and worse post-stroke recovery [9,10]. Understanding the genetic underpinnings of BBB dysfunction may provide valuable insights into stroke pathogenesis and pave the way for novel therapeutic strategies.
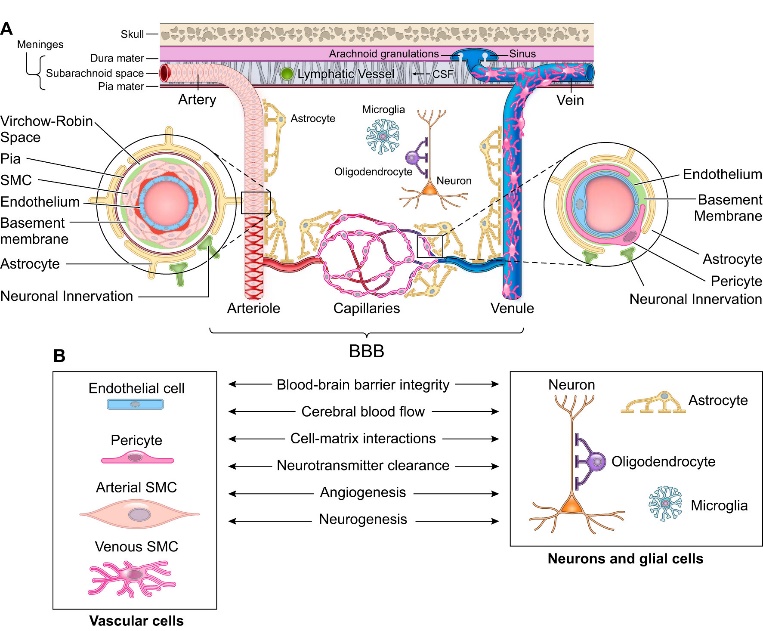
Figure1: The Neurovascular Unit
This review aims to explore the genetic mechanisms governing BBB integrity before and after ischemic stroke. We will discuss key genes involved in BBB maintenance, their roles in stroke pathophysiology, and the potential for genetic interventions to improve stroke outcomes. By synthesizing current knowledge, this review seeks to highlight the importance of genetic research in advancing our understanding of ischemic stroke and developing targeted therapies.
The Neurovascular Unit and BBB Structure
The neurovascular unit (NVU) is a complex structure comprising vascular cells, glial cells, and neurons, all of which contribute to BBB integrity and function (Figure. 1). At the capillary level, endothelial cells form tight junctions, which are critical for maintaining the BBB's selective permeability. Pericytes and astrocytes provide structural and functional support, while neurons regulate blood flow and BBB function through neurovascular coupling [11].
The neurovascular unit comprises vascular cells including endothelial cells and mural cells such as pericytes on brain capillaries, venules, and precapillary arterioles; vascular smooth muscle cells (SMC) on arterioles, small arteries, and veins; glial cells such as astrocytes, microglia, and olidogendrocytes; and neurons. Molecular expression patterns in endothelial and mural cells vary at different levels of vascular tree creating arterio-capillary-venous heterogeneity (zonation). At the level of penetrating arteries (left inset), endothelial cells form the inner layer of the vessel wall. The basement membrane separates endothelium from 1 to 3 layers of SMCs that are enveloped by pia. The Virchow-Robin space is between the pia and the glia limitans formed by astrocytic endfeet. At the arteriolar level, SMCs were reduced to a single layer, whereas the endothelial layer displays a continuity with the endothelium of penetrating arteries and capillaries. At the capillary level (right inset), pericytes and endothelial cells share a basement membrane and exhibit different types of cellular connections. Both the arteriolar and capillary vessel wall is covered by astrocytic endfeet. SMCs, pericytes, and astrocytes all have neuronal innervation. The blood-brain barrier (BBB) is centrally positioned within the neurovascular unit and is formed by a monolayer of tightly sealed endothelial cells extending along the vascular tree and expressing low paracellular and transcellular permeability at the level of brain capillaries and along arteriovenous axis. B: different cells of the neurovascular unit regulate BBB integrity, cerebral blood flow, extracellular matrix interactions, and neurotransmitter clearance and participate in angiogenesis and neurogenesis [11].
Genes Involved in BBB Integrity and Ischemic Stroke
The integrity of the BBB is regulated by a complex network of genes that encode proteins involved in tight junctions, transporters, inflammatory responses, and repair mechanisms. Dysregulation of these genes contributes to BBB disruption during ischemic stroke and influences stroke outcomes. Below, we discuss key genes and their roles in BBB maintenance and dysfunction.
Tight Junction Proteins
Tight junctions are critical for maintaining the selective permeability of the BBB. Genes encoding tight junction proteins are often dysregulated during ischemic stroke, leading to increased BBB permeability.
CLDN5 (Claudin-5)
- Role: A major component of tight junctions, regulating paracellular permeability
- Impact in Stroke: Downregulation of CLDN5 during stroke exacerbates BBB leakage and edema formation [11]
OCLN (Occludin)
- Role: Stabilizes tight junctions and regulates barrier function
- Impact in Stroke: Reduced OCLN expression is associated with increased infarct size and poor recovery [12]
TJP1 (ZO-1, Zonula Occludens-1)
- Role: Scaffold protein linking tight junctions to the actin cytoskeleton
- Impact in Stroke: Disruption of TJP1 leads to loss of BBB integrity and neuroinflammation [13]
Transporters and Channels
Transporters and channels regulate the movement of molecules across the BBB. Genetic variants in these genes can alter BBB function and influence stroke susceptibility.
ABCB1 (P-glycoprotein)
- Role: Efflux transporter that limits the entry of toxins and drugs into the brain
- Impact in Stroke: Polymorphisms in ABCB1 are associated with increased stroke risk and worse outcomes [14]
SLC2A1 (GLUT1, Glucose Transporter 1)
- Role: Facilitates glucose transport across the BBB
- Impact in Stroke: Reduced SLC2A1 expression during stroke leads to energy failure and neuronal damage [15]
AQP4 (Aquaporin-4)
- Role: Water channel involved in brain edema regulation
- Impact in Stroke: Upregulation of AQP4 exacerbates post-stroke edema, while its inhibition may have protective effects [16]
Inflammatory and Immune-Related Genes
Inflammation plays a central role in BBB disruption during ischemic stroke. Genes encoding cytokines, adhesion molecules, and immune regulators are key players.
IL-6 (Interleukin-6)
- Role: Pro-inflammatory cytokine that promotes BBB leakage
- Impact in Stroke: Elevated IL-6 levels correlate with worse stroke outcomes [17]
TNF-α (Tumor Necrosis Factor-alpha)
- Role: Induces endothelial cell activation and tight junction degradation
- Impact in Stroke: TNF-α inhibition reduces BBB disruption and infarct size [18]
ICAM-1 (Intercellular Adhesion Molecule 1)
- Role: Mediates leukocyte adhesion and infiltration into the brain.
- Impact in Stroke: Increased ICAM-1 expression contributes to neuroinflammation and BBB damage [19].
Matrix Metalloproteinases (MMPs)
MMPs are enzymes that degrade extracellular matrix components and tight junction proteins, leading to BBB breakdown.
MMP-9 (Matrix Metalloproteinase-9)
- Role: Degrades tight junction proteins and basement membrane components
- Impact in Stroke: Elevated MMP-9 levels are associated with hemorrhagic transformation and poor outcomes [20]
MMP-2 (Matrix Metalloproteinase-2)
- Role: Involved in early BBB disruption and angiogenesis
- Impact in Stroke: MMP-2 inhibition protects BBB integrity in experimental stroke models [21]
Growth Factors and Angiogenic Genes
Growth factors play a dual role in BBB repair and damage after ischemic stroke.
VEGF (Vascular Endothelial Growth Factor)
- Role: Promotes angiogenesis but also increases BBB permeability
- Impact in Stroke: VEGF overexpression exacerbates edema, while its inhibition enhances recovery [22].
BDNF (Brain-Derived Neurotrophic Factor)
- Role: Supports neuronal survival and BBB repair
- Impact in Stroke: BDNF polymorphisms are linked to stroke susceptibility and recovery [23]
Angiopoietins (ANGPT1, ANGPT2)
- Role: Regulate endothelial cell stability and vascular remodeling
- Impact in Stroke: ANGPT1 protects BBB integrity, while ANGPT2 promotes leakage [24]
Animal Models and Genetic Studies in BBB Integrity and Ischemic Stroke
Animal models have been instrumental in elucidating the molecular and cellular mechanisms underlying BBB dysfunction in ischemic stroke. Genetic studies, including transgenic and knockout models, provide insights into the roles of specific genes in BBB maintenance, disruption, and repair. This section discusses commonly used animal models and key genetic studies in the field.
Commonly Used Animal Models
Animal models of ischemic stroke mimic human stroke pathophysiology and allow for the study of BBB integrity in a controlled environment.
Middle Cerebral Artery Occlusion (MCAO)
- Description: A widely used model that involves transient or permanent occlusion of the middle cerebral artery, mimicking focal cerebral ischemia
- Advantages: Reproducible, mimics human stroke, and allows for the study of BBB disruption and repair over time
- BBB Studies: Used to investigate the roles of tight junction proteins, MMPs, and inflammatory mediators in BBB breakdown [25]
Photothrombotic Stroke Model
- Description: Involves the induction of focal ischemia by light activation of a photosensitive dye, causing endothelial damage and thrombosis
- Advantages: Precise control over infarct location and size
- BBB Studies: Useful for studying the effects of endothelial injury and oxidative stress on BBB integrity [26].
Global Ischemia Models
- Description: Mimics conditions such as cardiac arrest, where blood flow to the entire brain is temporarily interrupted
- Advantages: Models diffuse BBB damage and global cerebral ischemia
- BBB Studies: Used to study the effects of hypoxia and reperfusion on BBB permeability [27]
Transgenic and Knockout Models
Genetic manipulation in animal models allows researchers to study the roles of specific genes in BBB integrity and ischemic stroke.
CLDN5 Knockout Mice
- Findings: CLDN5 knockout mice exhibit increased BBB permeability and susceptibility to ischemic injury, highlighting the importance of claudin-5 in maintaining BBB integrity [28]
- Implications: Supports the role of tight junction proteins in stroke pathophysiology
MMP-9 Knockout Mice
- Findings: MMP-9 knockout mice show reduced BBB disruption and infarct size after stroke, demonstrating the role of MMP-9 in BBB breakdown [29]
- Implications: Suggests MMP-9 as a potential therapeutic target for stroke
VEGF Overexpression Models
- Findings: Overexpression of VEGF in mice leads to increased BBB permeability and edema, but also promotes angiogenesis and repair [30]
- Implications: Highlights the dual role of VEGF in stroke and the need for targeted therapies
Table 1: Summary of Key Genes and Their Roles
|
Gene |
Function |
Impact in Ischemic Stroke |
|
CLDN5 |
Tight junction protein |
Downregulation → BBB leakage |
|
ABCB1 |
Efflux transporter |
Polymorphisms → increased stroke risk |
|
MMP-9 |
Extracellular matrix degradation |
Elevated levels → hemorrhagic transformation |
|
VEGF |
Angiogenesis,vascular permeability |
Overexpression → edema |
|
IL-6 |
Pro-inflammatory cytokine |
Elevated levels → worse outcomes |
IL-6 Knockout Mice
- Findings: IL-6 knockout mice exhibit reduced neuroinflammation and BBB damage after stroke, underscoring the role of IL-6 in post-stroke inflammation [31]
- Implications: Supports the development of anti-inflammatory therapies for stroke
Genetic Studies in Stroke
Genetic studies in animal models and humans have identified key genes associated with BBB integrity and stroke outcomes.
Genome-Wide Association Studies (GWAS)
- Findings: GWAS in human populations have identified genetic variants associated with stroke risk and BBB dysfunction, such as polymorphisms in MMP-9, ABCB1, and CLDN5 [32]
- Implications: Provides insights into the genetic basis of stroke susceptibility and BBB regulation
CRISPR/Cas9 Gene Editing
- Applications: Used to create precise genetic modifications in animal models, such as knockouts or point mutations in BBB-related genes
- Example: CRISPR-mediated knockout of AQP4 in mice reduces post-stroke edema and improves outcomes [33]
Transcriptomic and Proteomic Studies
- Findings: High-throughput studies have identified gene expression changes in endothelial cells, astrocytes, and pericytes during stroke
- Example: Upregulation of ICAM-1 and VCAM-1 in endothelial cells during stroke highlights their role in leukocyte infiltration and BBB disruption [34]
Therapeutic Implications
Animal models and genetic studies have paved the way for the development of targeted therapies for stroke.
Gene Therapy
- Example: Delivery of VEGF or BDNF genes to promote angiogenesis and neuroprotection in stroke models [35].
Pharmacogenomics
- Example: Use of genetic information to personalize stroke treatments, such as selecting patients for MMP inhibitors based on MMP-9 polymorphisms [36]
Stem Cell Therapy
- Example: Transplantation of genetically modified stem cells to enhance BBB repair and neuroregeneration [37]
Impact of Ischemia on Gene Regulation
Ischemia induces profound changes in gene expression, affecting pathways involved in inflammation, oxidative stress, cell survival, and repair. These changes occur in various cell types, including endothelial cells, neurons, astrocytes, and microglia. Understanding the regulatory mechanisms behind these changes is crucial for developing targeted therapies for ischemic stroke.
Immediate Early Response Genes
Ischemia rapidly activates immediate early response genes (IEGs), which play a role in the initial cellular response to stress.
- Examples: FOS, JUN, EGR1
- Functions: Regulate transcription of downstream genes involved in inflammation, apoptosis, and repair.
- Impact: Activation of IEGs contributes to both protective and detrimental responses, depending on the context [35]
Inflammatory and Immune-Related Genes
Ischemia triggers a robust inflammatory response, leading to the upregulation of pro-inflammatory cytokines, chemokines, and adhesion molecules.
Key Genes
- IL-6 (Interleukin-6): Promotes inflammation and BBB disruption
- TNF-α (Tumor Necrosis Factor-alpha): Induces endothelial cell activation and apoptosis
- ICAM-1 (Intercellular Adhesion Molecule 1): Mediates leukocyte adhesion and infiltration
- Regulation: These genes are upregulated through activation of transcription factors such as NF-κB and STAT3 [36]
- Impact: Sustained inflammation exacerbates BBB damage and neuronal injury
Oxidative Stress-Related Genes
Ischemia-reperfusion injury generates reactive oxygen species (ROS), leading to oxidative stress and the activation of stress-responsive genes.
Key Genes
- SOD1 (Superoxide Dismutase 1): Antioxidant enzyme that neutralizes ROS
- HMOX1 (Heme Oxygenase 1): Protects against oxidative stress by degrading heme
- Nrf2 (Nuclear factor erythroid 2–related factor 2): Master regulator of antioxidant responses
- Regulation: Nrf2 activates the expression of antioxidant genes in response to oxidative stress [37]
- Impact: Dysregulation of these genes contributes to neuronal damage and BBB disruption
Apoptosis and Cell Survival Genes
Ischemia activates both pro-apoptotic and anti-apoptotic pathways, determining cell fate.
- Pro-Apoptotic Genes: BAX, CASP3 (Caspase-3): Promote programmed cell death
- Anti-Apoptotic Genes: BCL-2, BCL-XL: Inhibit apoptosis and promote cell survival
- Regulation: The balance between pro- and anti-apoptotic genes is influenced by transcription factors such as p53 and HIF-1α [38]
- Impact: Excessive apoptosis contributes to neuronal loss, while activation of survival pathways may promote recovery
Hypoxia-Inducible Factors (HIFs)
Hypoxia-inducible factors are key regulators of the cellular response to low oxygen levels.
Key Genes
- HIF-1α: Regulates genes involved in angiogenesis, metabolism, and survival
- VEGF (Vascular Endothelial Growth Factor): Promotes angiogenesis but also increases BBB permeability
- Regulation: HIF-1α is stabilized under hypoxic conditions and activates target genes such as VEGF and GLUT1 [39]
- Impact: HIF-1α activation has dual roles in promoting both tissue repair and BBB disruption
Matrix Metalloproteinases (MMPs)
MMPs are upregulated during ischemia and contribute to BBB breakdown.
Key Genes
- MMP-9: Degrades tight junction proteins and extracellular matrix components
- MMP-2: Involved in early BBB disruption and angiogenesis
- Regulation: MMP expression is regulated by cytokines, ROS, and transcription factors such as NF-κB [39]
- Impact: MMP-mediated BBB disruption exacerbates brain injury and edema
Repair and Regeneration Genes
Ischemia also activates genes involved in tissue repair and regeneration.
Key Genes
- BDNF (Brain-Derived Neurotrophic Factor): Promotes neuronal survival and plasticity
- ANGPT1 (Angiopoietin-1): Stabilizes blood vessels and supports BBB repair
- GFAP (Glial Fibrillary Acidic Protein): Marker of astrocyte activation and glial scar formation
- Regulation: These genes are upregulated during the subacute and chronic phases of stroke [40]
- Impact: Activation of repair mechanisms is essential for long-term recovery but may also contribute to glial scar formation
MMP-9 Extracellular matrix degradation, NF-κB, ROS Contributes to BBB breakdown
Targeting Genetic Pathways
Gene Therapy: Gene therapy involves the delivery of genetic material to cells to modify gene expression or correct genetic defects. In the context of ischemic stroke, gene therapy can be used to enhance neuroprotection, promote BBB repair, and support tissue regeneration.
Approaches
- Viral Vectors: Adeno-associated viruses (AAVs) and lentiviruses are commonly used to deliver therapeutic genes to the brain
- Non-Viral Methods: Nanoparticles and liposomes can be used for gene delivery, offering lower immunogenicity and easier production
Examples
- VEGF Delivery: Overexpression of VEGF promotes angiogenesis and neuroprotection but must be carefully controlled to avoid exacerbating BBB leakage [1].
- BDNF Delivery: BDNF gene therapy supports neuronal survival and plasticity, improving functional recovery after stroke [2].
Challenges
- Ensuring targeted delivery to affected brain regions
- Avoiding off-target effects and immune responses
CRISPR-Based Approaches
CRISPR-Cas9 technology enables precise genome editing, offering the potential to correct genetic mutations or modulate gene expression in ischemic stroke.
Applications
- Gene Knockout: Inactivation of detrimental genes (e.g., MMP-9) to reduce BBB disruption and neuroinflammation
- Gene Activation: Upregulation of protective genes (e.g., Nrf2, HMOX1) to enhance antioxidant responses and cell survival
- Base Editing: Correction of single-nucleotide polymorphisms (SNPs) associated with stroke risk (e.g., ABCB1 variants) [18]
Examples
- MMP-9 Knockout: CRISPR-mediated knockout of MMP-9 in animal models reduces BBB leakage and infarct size [22]
- Nrf2 Activation: CRISPR activation of Nrf2 enhances antioxidant defenses and reduces oxidative stress [16]
Challenges
- Ensuring efficient and safe delivery to the brain
- Addressing ethical concerns and potential off-target effects
Pharmacogenomics and Personalized Medicine
Pharmacogenomics involves tailoring treatments based on an individual's genetic profile. This approach can optimize drug efficacy and minimize adverse effects in stroke patients.
Key Areas
- Stroke Risk Prediction: Identifying genetic variants (e.g., MMP-9, ABCB1) associated with increased stroke risk to guide preventive strategies
- Drug Response: Using genetic information to predict responses to thrombolytics (e.g., tPA) and antiplatelet agents (e.g., clopidogrel)
- BBB-Targeted Therapies: Developing drugs that modulate BBB-related genes (e.g., CLDN5, VEGF) to enhance repair and reduce damage
Examples
- tPA Pharmacogenomics: Genetic variants in PLAT (tissue plasminogen activator) and SERPINE1 (PAI-1) influence tPA efficacy and bleeding risk [26]
- Clopidogrel Response: Variants in CYP2C19 affect clopidogrel metabolism, guiding the use of alternative antiplatelet agents [37]
Challenges
- Integrating genetic testing into clinical practice
- Addressing the complexity of polygenic traits and gene-environment interactions
Neuroprotective Strategies
Targeting genetic pathways can also enhance neuroprotection and BBB repair after ischemic stroke.
Examples
- Anti-Inflammatory Therapies: Targeting genes such as IL-6 and TNF-α to reduce neuroinflammation and BBB disruption
- Antioxidant Therapies: Activating Nrf2-mediated pathways to combat oxidative stress
- Angiogenic Therapies: Modulating VEGF and ANGPT1 expression to promote vascular repair while minimizing edema
Challenges and Future Directions
While targeting genetic pathways holds great promise, several challenges must be addressed:
- Delivery Systems: Developing safe and efficient methods for delivering gene therapies and CRISPR components to the brain
- Ethical Considerations: Ensuring responsible use of genome editing technologies
- Personalized Approaches: Integrating multi-omics data (genomics, transcriptomics, proteomics) to develop tailored therapies
- Clinical Translation: Moving from preclinical studies to clinical trials and routine clinical practice
CONCLUSIONS
The blood-brain barrier (BBB) plays a critical role in maintaining brain homeostasis, and its integrity is profoundly affected by ischemic stroke. Understanding the genetic mechanisms underlying BBB dysfunction and repair is essential for developing effective therapeutic strategies. This review has highlighted the key genes and pathways involved in BBB regulation, including tight junction proteins, transporters, inflammatory mediators, and growth factors. We have also explored the impact of ischemia on gene regulation, emphasizing the roles of oxidative stress, inflammation, and apoptosis in stroke pathophysiology.
Animal models and genetic studies have provided invaluable insights into the molecular mechanisms of BBB disruption and repair. These studies have identified potential therapeutic targets, such as MMP-9, VEGF, and Nrf2, which can be modulated to enhance neuroprotection and promote recovery. Advances in gene therapy, CRISPR-based approaches, and pharmacogenomics offer promising avenues for personalized stroke treatment, with the potential to improve outcomes and reduce the burden of stroke-related disability.
However, significant challenges remain, including the need for efficient and safe delivery systems, the integration of multi-omics data, and the translation of preclinical findings into clinical practice. Future research should focus on addressing these challenges and exploring the complex interplay between genetic, epigenetic, and environmental factors in stroke pathogenesis.
In conclusion, targeting genetic pathways represents a transformative approach to stroke treatment, with the potential to revolutionize patient care. By leveraging the power of genetics and genomics, we can move closer to achieving precision medicine for ischemic stroke, ultimately improving the lives of millions of affected individuals worldwide.
REFERENCES
- Zhang, R.L. et al. "Anti-intercellular adhesion molecule-1 antibody reduces ischemic cell damage after transient but not permanent middle cerebral artery occlusion in the Wistar rat." Stroke, 1995.
- Frijns, C.J. and Kappelle, L.J. "Inflammatory cell adhesion molecules in ischemic cerebrovascular disease." Stroke, 2002.
- Wang, L. et al. "ICAM-1 deficiency reduces blood-brain barrier damage in experimental ischemic stroke." Neurological Research, 2013.
- Abbott, N.J. et al. "Structure and function of the blood-brain barrier." Neurobiology of Disease, 2010.
- Abdullahi, W. et al. "Blood-brain barrier dysfunction in ischemic stroke: targeting tight junctions and transporters for vascular protection." American Journal of Physiology Cell Physiology, 2018.
- Campbell, B.C. et al. "Ischaemic stroke." Nature Reviews Disease Primers, 2019.
- Chen, J. et al. "CRISPR-Cas9-mediated gene editing for ischemic stroke: promises and challenges." Molecular Neurobiology, 2023.
- Chen, W. et al. "CRISPR-Cas9 gene editing for neurological disorders: progress and challenges." Molecular Therapy, 2020.
- Daneman, R. and Prat, A. "The blood-brain barrier." Cold Spring Harbor Perspectives in Biology, 2015.
- Goulay, R. et al. "Subarachnoid hemorrhage impairs cerebrospinal fluid circulation." Stroke, 2020.
- Jiang, X. et al. "Blood-brain barrier dysfunction and recovery after ischemic stroke." Progress in Neurobiology, 2021.
- Jickling, G.C. et al. "Targeting neutrophils in ischemic stroke." Journal of Cerebral Blood Flow and Metabolism, 2015.
- Kadry, H. et al. "A blood-brain barrier overview on structure, function, impairment and biomarkers." Fluids and Barriers of the CNS, 2020.
- Armulik, A. et al. "Pericytes regulate the blood-brain barrier." Nature, 2010.
- Liu, J. et al. "Matrix metalloproteinase-2-mediated occludin degradation and claudin-5 redistribution in stroke." Journal of Neuroscience, 2012.
- Armulik, A. et al. "Pericytes regulate the blood-brain barrier." Nature, 2010.
- Liu, J. et al. "Matrix metalloproteinase-2-mediated occludin degradation and claudin-5 redistribution in stroke." Journal of Neuroscience, 2012.
- Löscher, W. and Potschka, H. "Role of drug efflux transporters in brain diseases." Progress in Neurobiology, 2005.
- Cordon-Cardo, C. et al. "Multidrug-resistance gene expression at blood-brain barrier sites." Proceedings of the National Academy of Sciences of the United States of America, 1989.
- Jablonski, M.R. et al. "Selective inhibition of ABCB1 protects brain endothelial cells." Fluids and Barriers of the CNS, 2012.
- Winkler, E.A. et al. "GLUT1 reductions exacerbate Alzheimer's disease dysfunction." Nature Neuroscience, 2015.
- Yu, S. et al. "Reduced cerebral glucose uptake in Alzheimer’s model." Journal of Molecular Neuroscience, 2014.
- Simpson, I.A. et al. "Supply and demand in cerebral energy metabolism." Journal of Cerebral Blood Flow and Metabolism, 2007.
- Armulik, A. et al. "Pericytes regulate the blood-brain barrier." Nature, 2010.
- Manley, G.T. et al. "Aquaporin-4 deletion reduces brain edema." Nature Medicine, 2000.
- Papadopoulos, M.C. and Verkman, A.S. "Aquaporin water channels in the nervous system." Nature Reviews Neuroscience, 2013.
- Fukuda, A.M. and Badaut, J. "Aquaporin 4 in cerebral edema and neuroinflammation." Journal of Neuroinflammation, 2012.
- Liu, J. et al. "Matrix metalloproteinase-2-mediated occludin degradation in stroke." Journal of Neuroscience, 2012.
- Li, T. et al. "Matrix metalloproteinases in blood-brain barrier dysfunction." Frontiers in Neuroscience, 2021.
- Liebner, S. et al. "Functional morphology of the blood-brain barrier." Acta Neuropathologica, 2021.
- Liu, J. et al. "Matrix metalloproteinase-2-mediated occludin degradation." Journal of Neuroscience, 2012.
- Liu, Y. et al. "Nanotechnology-based drug delivery systems for ischemic stroke." Advanced Drug Delivery Reviews, 2023.
- Smith, C.J. et al. "Interleukin-6 and inflammation markers in ischemic stroke." BMC Neurology, 2004.
- Zaidan, S.R. "Interaction studies of insulin degrading enzyme with membrane." Frontiers in Health Informatics, 2024.
- Lasek-Bal, A. et al. "Importance of IL-6, IL-8, and TNF-α in stroke severity." Disease Markers, 2016.
- Malik, R. et al. "Genome-wide meta-analysis identifies loci associated with stroke." Annals of Neurology, 2018.
- Obermeier, B. et al. "Development and disruption of the blood-brain barrier." Nature Medicine, 2013.
- Profaci, C.P. et al. "The blood-brain barrier in health and disease." Journal of Experimental Medicine, 2020.
- Shi, Y. et al. "Endothelial cytoskeletal reorganization and blood-brain barrier disruption." Nature Communications, 2022.
- Sweeney, M.D. et al. "Blood-brain barrier breakdown in neurodegenerative disorders." Nature Reviews Neurology, 2020.

